 
The CIP algorithm in ChemDoodle is validated against the test suite provided by Hanson et. Stereochemical features in your structures will be assigned "R", "S", "E", "Z", "M" and "P" descriptors. to remove any ambiguities and describe a completely consistent system for CIP assignments.ĬhemDoodle implements all 6 current CIP rules as well as auxilliary desciptors and mancude ring support. The most recent CIP rules from IUPAC were then algorithmically analyzed and standarized by Hanson et al. These rules were adopted by IUPAC for naming standards and fully described in the Blue books. While flawed, they have seen many revisions over the decades and were clarified by the work of Paulina Mata. A lot of times, the reaction conditions such as the temperature, pressure, catalyst etc are indicated above and/or below the arrow in the equation.The CIP rules have long been the standard for describing configurations of stereochemical features in a molecule. Physical states of the reactants and products are usually not specified in the equation unless necessary. Note – For H 2O, the symbol (g) is used to indicate that here, water has been used in the form of steam. The balanced equation above now becomes –ģFe(s) + 4H 2O(g) → Fe 3O 4 (s) + 4H 2(g) If the reactant or product exists as a solution in water, it is written as aqueous (aq). The phases of the reactants and products are mentioned using notations such as (g) for gaseous, (l) for liquid, (s) for solid and (aq) for aqueous. This makes the balanced equation more informative. The last step is to write the physical states of the reactants and products. As we make trials to balance the equation using smallest whole numbers as coefficients, this method is called the ‘hit and trial method’ of balancing equations. The number of atoms of each element on both sides is now equal, therefore this equation is now a balanced equation. Now, we count the atoms of each element on both sides of the equation to the check the correctness of the equation 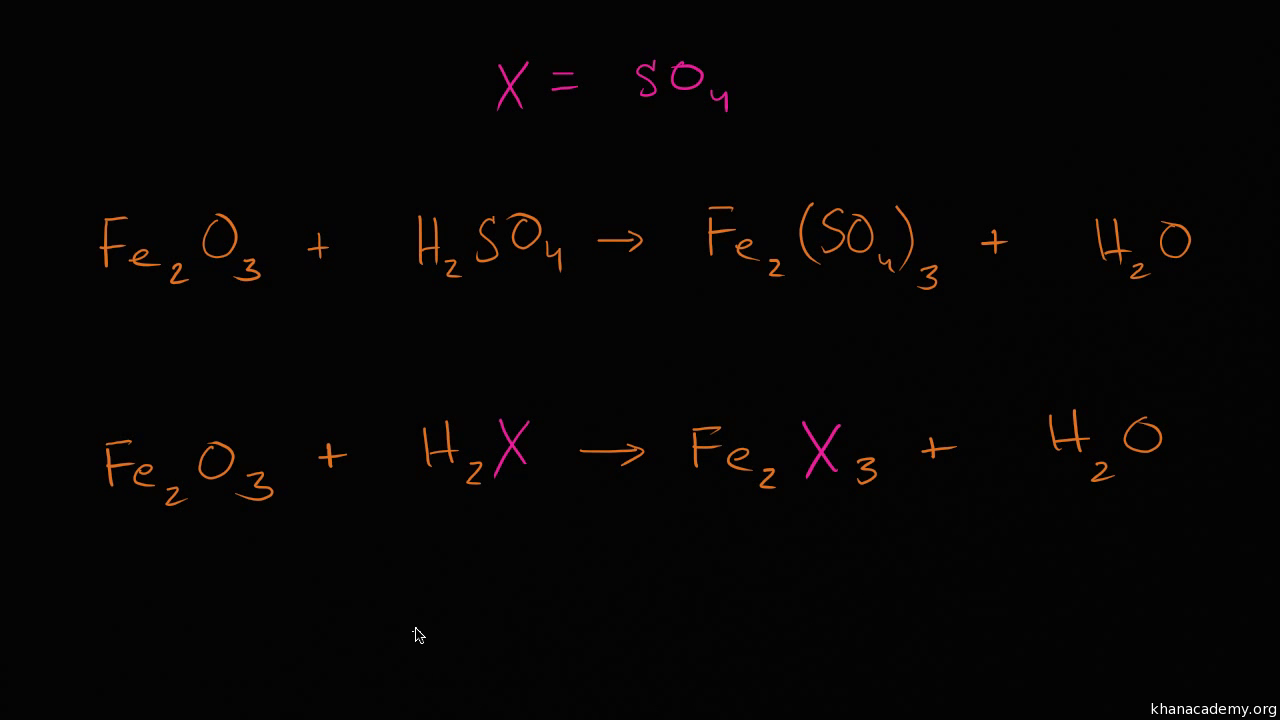 
Therefore, we add 3 as a coefficient to Fe on the LHS – Let’s try to balance that – Atoms of iron Now, you will find that only one element is left unbalanced i.e. Therefore, we add 4 as a coefficient to H 2 on the RHS. Now, let’s balance the hydrogen atoms in the equation – Atoms of hydrogen Now, the partly balanced equation looks like this – For example, to balance the number of oxygen atoms, we can add 4 as a coefficient to get 4H 2O and not H 2O 4 or (H 2O) 4. Remember – To equalize the number of atoms in a reaction, you cannot change the formulae of the compounds or the elements in the reaction. Let’s try to balance the oxygen atoms – Atoms of oxygen As you can see, there are 4 oxygen atoms on the RHS, whereas there is only one oxygen atom on the LHS. Now, in this compound select the element with the maximum number of atoms i.e. To balance the above equation, let’s start with the compound with the maximum number of atoms i.e. Fe 3O 4. List the number of atoms of each element on either side of the arrow in the equation – Element We can balance this equation using the following steps – Step Iįirst, draw boxes around each formula without changing anything inside the boxes.īrowse more Topics under Chemical Reactions And Equations Therefore, this is an ‘unbalanced’ equation. If you notice, the number of atoms of each element are not the same on both sides of the arrow. Therefore, this chemical equation is a ‘balanced equation’. Now, let us look at the number of atoms of each element on LHS and RHS – ElementĪs you can see, the number of atoms of each element on LHS and RHS are the same. Zinc + Sulphuric acid → Zinc sulfate + Hydrogen For example, let’s look at the following chemical equation – The number of atoms of each element remains the same, before and after a chemical reaction. This means that the total mass of elements in the products of a chemical reaction has to be equal to the total mass of the elements in the reactants. This law applies to a chemical reaction too. As you have already learned earlier, mass can neither be created nor destroyed. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
